| Balloon Type | Rapid Exchange (Rx) |
| Compliance Type | Semi-Compliant |
| Balloon Folding | Tri-Fold |
| Total Working Length | 142 |
| Stent Lengths (in mm) | 8mm to 38 mm ( 8,13,18,23,28,33,38) |
| Stent Diameters (in mm) | 2.50 mm to 4.00 mm (2.50,2.75,3.00,3.25,3.50,3.75 & 4.00 mm) |
| Distal Shaft Diameter | 2.6 Fr |
| Proximal Shaft Diameter | 1.9 Fr |
| Balloon Nominal Pressure (N/P) | 8 atm |
| Balloon Rated Burst Pressure (RBP) | 16 atm |
|
Crossing Profile 2.50/2.75/3.00 3.25/ 3.50/4.00 |
Profile (mm) < 1.10 < 1.15 |
| Balloon Material | Nylon |
| Marker Material | Platinum-Iridium |
| Radiopaque Markers | Dual Markers |
| Tip Length | 4mm |
| Tip Entry Profile | 0.019” |
| Distal Coating | Hydrophilic |
| Minimum Guiding Catheter ID | 5 Fr |
| Maximum Guide Wire Compatibility | 0.014” |
| Inflation Time | ≤ 10 sec |
| Deflation Time | ≤ 25 sec |
| Shelf Life | 18 months |
| STENT SPECIFICATIONS | |
| Design | Hybrid |
| Material | L 605 Cobalt Chromium Alloy |
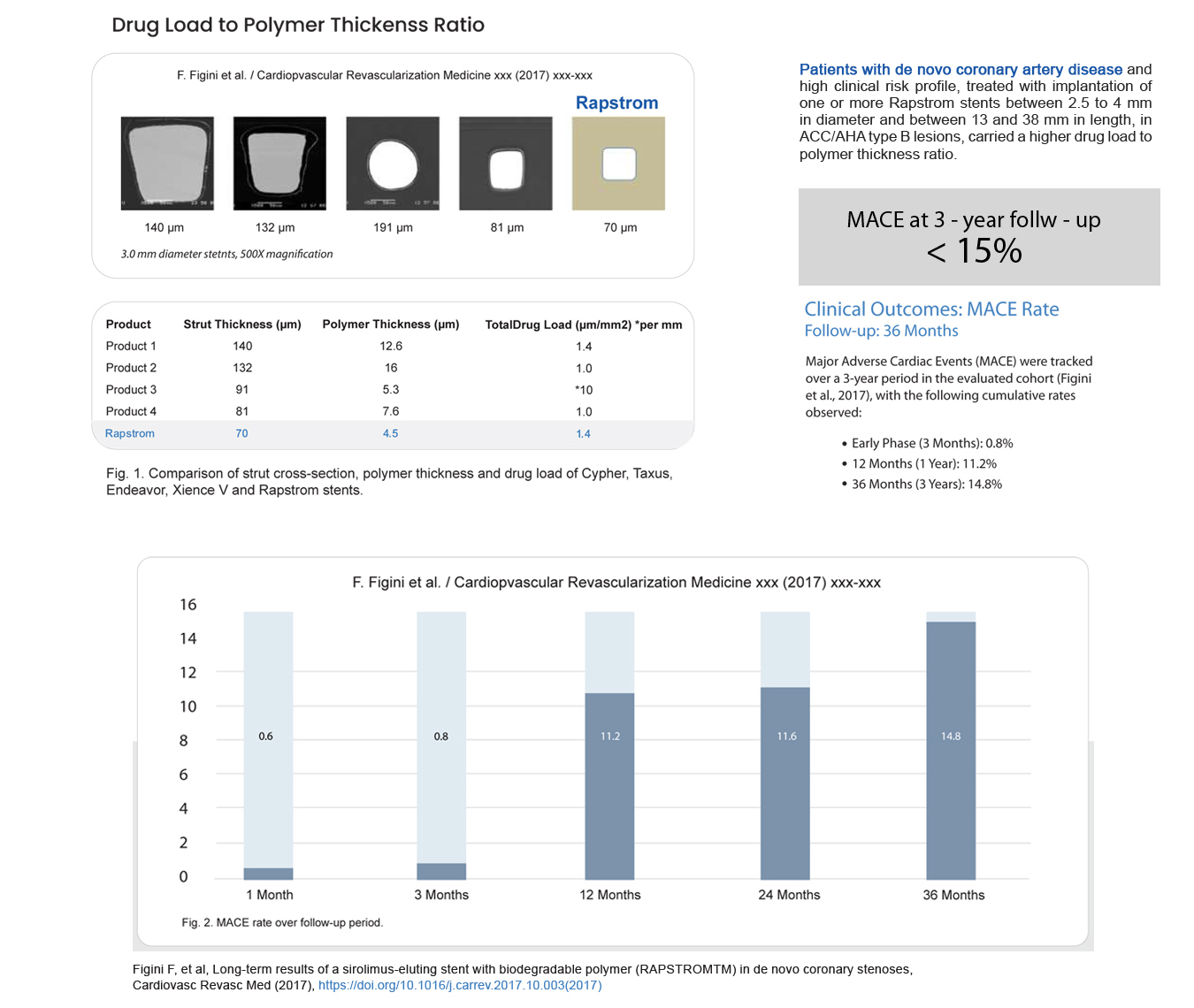
| Strut Thickness | 70 μm (0.070 mm) |
| Strut Width | 100 μm (0.10 mm) |
| Radial Strength | 0.8 N/mm |
| Maximum Expansion Profile | ≤ 4 atm beyond RBP pressure 0.1 – 0.2 mm |
| Drug | Sirolimus |
| Drug Dose | 1.4µg/mm² (Sirolimus per square mm of stent surface area) |
|
Drug Elution Profile Day 1 Day 7 Total Drug on Stent |
25% – 45% of label claim 50% – 80% of label claim |
| Polymer | PLLA-PLGA |
| Polymer | 6 months (from time of implantation) |
| Mean Recoil | <5% |
| Mean Foreshortening | <3% |